Photocatalytic evaluation of supported ilmenite on clay substrates for degradation of organic dyes
DOI:
https://doi.org/10.47566/2026_syv39_1-260401Keywords:
Photocatalysis, Ilmenite, Degradation, DyesAbstract
Titanium dioxide (TiO2) is widely used as a photocatalyst for the degradation of organic pollutants in water. However, to improve its absorption in the visible range and enhance the potential of photocatalysis for water treatment, the incorporation of Fe into TiO2 or the use of its natural mineral form, such as ilmenite (Fe2+Ti4+O3), has been widely studied. However, the effects of the different compositions in natural ilmenite minerals on the photocatalytic degradation of dyes has not been revised. In this study, the photodegradation of methylene blue (MB) and methyl orange (MO) dyes was studied using five different ilmenite minerals under UV and visible light irradiation. For the case of visible light photocatalysis a pilot scale reactor was tested. The ilmenites were characterized by diffraction (XRD), X-ray fluorescence (XRF) and diffuse reflectance spectroscopy (DRS). Crystalline phases included rutile, pseudorutile, and ilmenite. The Fe/Ti ratio in the ilmenites was between 1:10 to 1:1, while V and Mn were found as minor impurities (0 to 2 wt% Mn, and 1.0 to
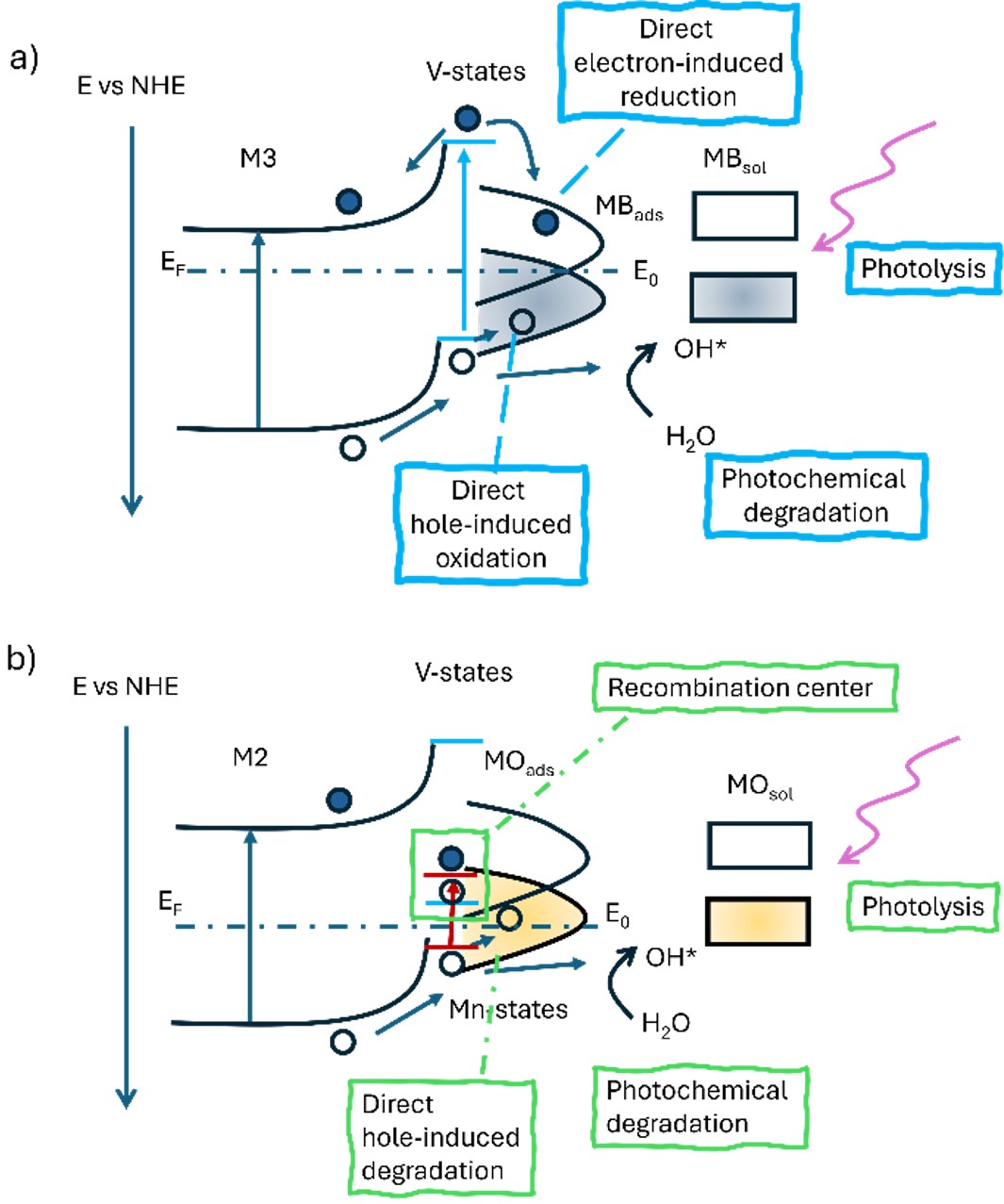
1.6 wt% V). Band gap energies vary with the Fe/Ti ratio from 2.18 to 2.83 eV. Photodegradation experiments were conducted with an initial dye concentration of 6 mg/L, over three cycles of 200 minutes, collecting samples at various time intervals. Under UV light, degradation percentages for MB and MO ranged from 48 to 90% and 26 to 74%, respectively, by the end of the third degradation cycle. Under visible light, samples with the higher Ti/Fe ratio (M3 and M5) achieved 61.10% and 57.73% degradation of MB, respectively. No degradation was observed for MO under visible light, while MB degradation was attributed to a sensitization effect on TiO2. The differences in photocatalytic efficiency between dyes were attributed to their molecular structures, to the Fe/Ti ratios and the V and Mn impurities in the ilmenites, although further work is required to differentiate between these effects. The work demonstrated the feasibility of using natural ilmenites for dye photodegradation, as well as to set a pilot scale reactor for visible light treatments.
References
[1]. L.I. Diaconu, C.I. Covaliu-Mierla, O. Paunescu, L.D. Covaliu, H. Iovu, G. Paraschiv, “Phytoremediation of Wastewater Containing Lead and Manganese Ions Using Algae”, Biology 12, 773 (2023).
https://doi.org/10.3390/biology12060773
[2]. S.H. Hashemi, M. Kaykhaii, Azo dyes: Sources, occurrence, toxicity, sampling, analysis, and their removal methods. In: Emerging Freshwater Pollutants, Eds.: T. Dalu, N.T. Tavengwa, (Elsevier, 2022) p. 267-287.
https://doi.org/10.1016/B978-0-12-822850-0.00013-2
[3]. S.N. Tan, M.L. Yuen, R.A. Ramli, “Photocatalysis of dyes: Operational parameters, mechanisms, and degradation pathway”, Green Anal. Chem. 12, 100230 (2025).
https://doi.org/10.1016/j.greeac.2025.100230
[4]. Y. Wang, C. Yang, Y. Liu, Y. Fan, F. Dang, Y. Qiu, H. Zhou, W. Wang, Y. Liu, “Solvothermal Synthesis of ZnO Nanoparticles for Photocatalytic Degradation of Methyl Orange and p-Nitrophenol”, Water 13, 3224 (2021).
https://doi.org/10.3390/w13223224
[5]. X. Zhu, J. Wang, D. Yang, J. Liu, L. He, M. Tang, W. Feng, X. Wu, “Fabrication, characterization and high photocatalytic activity of Ag–ZnO heterojunctions under UV-visible light”, RSC Adv. 11, 27257 (2021).
https://doi.org/10.1039/D1RA05060E
[6]. K.M. Mohamed, J.J. Benitto, J.J. Vijaya, M. Bououdina, “Recent Advances in ZnO-Based Nanostructures for the Photocatalytic Degradation of Hazardous, Non-Biodegradable Medicines”, Crystals 13, 329 (2023).
https://doi.org/10.3390/cryst13020329
[7]. C.B. Anucha, I. Altin, E. Bacaksiz, V.N. Stathopoulos, “Titanium dioxide (TiO2)-based photocatalyst materials activity enhancement for contaminants of emerging concern (CECs) degradation: In the light of modification strategies”, Chem. Eng. J. Adv. 10, 100262 (2022).
https://doi.org/10.1016/j.ceja.2022.100262
[8]. K. Nagaraj, S. Radha, C.G. Deepa, K. Raja, V. Umapathy, N. Prakash Badgujar, N.M. Parekh, T. Manimegalai, L. Archana Devi, C. Uthra, “Photocatalytic advancements and applications of titanium dioxide (TiO2): Progress in biomedical, environmental, and energy sustainability”, Next Research 2, 100180 (2025).
https://doi.org/10.1016/j.nexres.2025.100180
[9]. H. Moradi, A. Eshaghi, S.R. Hosseini, K. Ghani, “Fabrication of Fe-doped TiO2 nanoparticles and investigation of photocatalytic decolorization of reactive red 198 under visible light irradiation”, Ultrason. Sonochem. 32, 314 (2016).
https://doi.org/10.1016/j.ultsonch.2016.03.025
[10]. C. Adán, J. Carbajo, A. Bahamonde, A. Martínez-Arias, “Phenol photodegradation with oxygen and hydrogen peroxide over TiO2 and Fe-doped TiO2”, Catal. Today 143, 247 (2009).
https://doi.org/10.1016/j.cattod.2008.10.003
[11]. R.B. Lee, J.C. Juan, C.W. Lai, K.M. Lee, “Ilmenite: Properties and photodegradation kinetic on Reactive Black 5 dye Ilmenite: Properties and photodegradation kinetic on Reactive Black 5 dye”, Chin. Chem. Lett. 28, 1613 (2017).
https://doi.org/10.1016/j.cclet.2017.03.006
[12]. E.Y. Fernando, D. Sarkar, C. Rodwihok, A. Satpathy, J. Zhang, R. Rahmati, R. Datta, C. Christodoulatos, M. Boufadel, S. Larson, Z. Zhang, “Unrefined and Milled Ilmenite as a Cost-Effective Photocatalyst for UV-Assisted Destruction and Mineralization of PFAS”, Materials 17, 3801 (2024).
https://doi.org/10.3390/ma17153801
[13]. R.A. Putri, S. Tursiloadi, E.F. Nurrahmah, A.R. Liandi, O. Arutanti, “Synthesis of TiO2-Based Photocatalyst from Indonesia Ilmenite Ore for Photodegradation of Eriochrome Black-T Dye”, Water Air Soil Pollut. 234, 113 (2023).
https://doi.org/10.1007/s11270-023-06584-2
[14]. M.E. Besheli, M. Heidari-Golafzani, T. Mohammadi, R. Rahimi, “Using an economic method to prepare TiO2 from natural ilmenite for photodegradable dye removal”, Results Chem. 7, 101297 (2024).
https://doi.org/10.1016/j.rechem.2023.101297
[15]. H.M. Ali, F.A. Roghabadi, V. Ahmadi, “Solid-supported photocatalysts for wastewater treatment: Supports contribution in the photocatalysis process”, Solar Energy 255, 99 (2023).
https://doi.org/10.1016/j.solener.2023.03.032
[16]. X. Li, U. Simon, M.F. Bekheet, A. Gurlo, “Mineral-Supported Photocatalysts: A Review of Materials, Mechanisms and Environmental Applications”, Energies 15, 5607 (2022).
https://doi.org/10.3390/en15155607
[17]. C. Guarneros Aguilar, C. Estrada Moreno, M. Pacio Castillo, F. Caballero-Briones, “Effect of calcination temperature on structure and thermoelectric properties of CuAlO2 powders”, J. Mater. Sci. 53, 1646 (2018).
https://doi.org/10.1007/s10853-017-1602-8
[18]. M. Taniguchi, Absorption coefficient spectrum of Methyl orange. Azo dyes; at PhotochemCAD™ (open in April 2026).
https://www.photochemcad.com/databases/common-compounds/azo-dyes/methyl-orange
[19]. S. Prahl, Tabulated Molar Extinction Coefficient for Methylene Blue in Water; at OMLC (open in April 2026).
https://omlc.org/spectra/mb/mb-water.html
[20]. O. Monfort, P. Petrisková, “Binary and Ternary Vanadium Oxides: General Overview, Physical Properties, and Photochemical Processes for Environmental Applications”, Processes 9, 214 (2021).
https://doi.org/10.3390/pr9020214
[21]. P. Pooseekheaw, W. Thongpan, A. Panthawan, E. Kantarak, W. Sroila, P. Singjai, “Porous V2O5/TiO2 Nanoheterostructure Films with Enhanced Visible-Light Photocatalytic Performance Prepared by the Sparking Method”, Molecules 25, 3327 (2020).
https://doi.org/10.3390/molecules25153327
[22]. Q. Luo, X.W. Li, Q.Z. Cai, Q.S. Yan, Z.H. Pan, “Preparation of narrow band gap V2O5/TiO2 composite films by micro-arc oxidation”, Int. J. Miner. Metall. Mater. 19, 1045 (2012).
https://doi.org/10.1007/s12613-012-0668-1
[23]. P.S. Nair, H. Rahman, J.A. Joseph, A. Norbert, S. Shaji, S. Tripathi, S.N. Jha, R.R. Philip, “Band gap engineering of TiO2 by Mn doping and the effect of p-TNT: Mn/n-MnO2 heterojunction on photocatalytic applications”, Appl. Phys. A 128, 1089 (2022).
https://doi.org/10.1007/s00339-022-06246-w
[24]. D. Hao, Q. Huang, W. Wei, X. Bai, B.J. Ni, “A reusable, separation-free and biodegradable calcium alginate/g-C3N4 microsphere for sustainable photocatalytic wastewater treatment”, J. Cleaner Prod. 314, 128033 (2021).
https://doi.org/10.1016/j.jclepro.2021.128033
[25]. H. Liang, Z. Wang, L. Liao, L. Chen, Z. Li, J. Feng, “High performance photocatalysts: Montmorillonite supported-nano TiO2 composites”, Optik 136, 44 (2017).
https://doi.org/10.1016/j.ijleo.2017.02.018
[26]. A. Kubiak, M. Jaruga, “Pilot-scale LED photocatalytic system utilizing 3D bioinspired sponge photocatalyst for the removal of amoxicillin, tetracycline, and ciprofloxacin from hospital wastewater”, Chem. Eng. J. 510, 161758 (2025).
https://doi.org/10.1016/j.cej.2025.161758
[27]. T. Lana Villarreal, R. Gomez, M. Neumann-Spallart, N. Alonso-Vante, P. Salvador, “Semiconductor Photooxidation of Pollutants Dissolved in Water: A Kinetic Model for Distinguishing between Direct and Indirect Interfacial Hole Transfer. I. Photoelectrochemical Experiments with Polycrystalline Anatase Electrodes under Current Doubling and Absence of Recombination”, J. Phys. Chem. B 108, 15172 (2004).
https://doi.org/10.1021/jp049447a
[28]. R. Rami, N. Rkhioui, R. Ahl Laamara, L.B. Drissi, “Electronic and magnetic properties of TiO2 (co)-doped with (V, Mn)”, Mater. Res. Express 4, 126513 (2017).
https://doi.org/10.1088/2053-1591/aa9e9f
[29]. D.M. Cañas-Martínez, G.H. Gauthier, J.A. Pedraza-Avella, “Photo-oxidative and photo-reductive capabilities of ilmenite-rich black sand concentrates using methyl orange as a probe molecule”, Photochem. Photobiol. Sci. 18, 912 (2019).
Downloads
Published
Issue
Section
License
Copyright (c) 2026 The authors; licensee SMCTSM, Mexico.

This work is licensed under a Creative Commons Attribution 4.0 International License.
©2026 by the authors; licensee SMCTSM, Mexico. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).





