Is sulfur the responsible for color of yellow Chiapas Amber?
DOI:
https://doi.org/10.47566/2021_syv34_1-211201Keywords:
Amber, XPS, composite material.Abstract
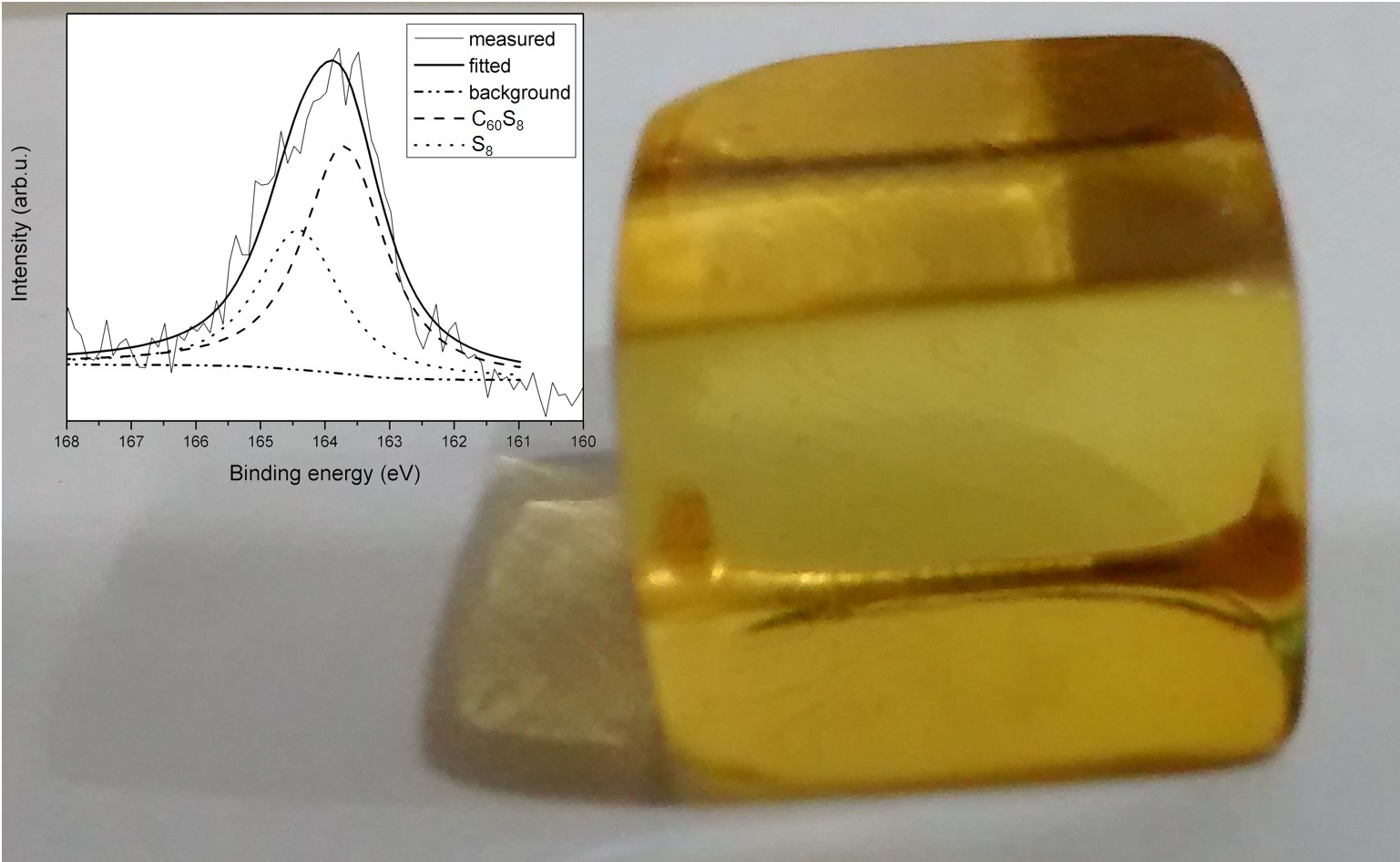
Amber is a fossilized natural resin found in specific areas of the world; its geographical origin is associated to a characteristic color. Amber all over the world share basic characteristics due to a common polymeric matrix; however, its color is associated to the environment where it was formed. In this work, based on a compositional analysis, it is proposed that yellow color of Mexican amber is originated from the sulfur physical and chemical interaction within its polymeric matrix. The effect of sulfur present in yellow Mexican amber is studied by employing X-ray photoelectron and UV-Vis spectroscopies. Results show that sulfur is incorporated inside the polymer matrix with two different chemical environments; one related with sulfur-carbon bonds and the other with sulfur-sulfur bonds. The optical transmission spectrum simulated considering amber as a composite material reproduce the scattering contribution observed experimentally.
References
. M. Riquelme, F. Méndez Acuña, Biodiversitas 127, 6, (2016).
https://bioteca.biodiversidad.gob.mx/janium/Documentos/12741.pdf
. J.H. Langenheim, Biology of Amber-Producing Trees: Focus on Case Studies of Hymenaea and Agathis. In: Amber, Resinite, and Fossil Resins, vol. 617, Eds. K.B. Anderson, J.C. Crelling, (American Chemical Society, 1996) pp. 1-31.
https://doi.org/10.1021/bk-1995-0617.ch001
. D.J. Clifford, P.G. Hatcher, Org. Geochem. 23, 407 (1995).
https://doi.org/10.1016/0146-6380(95)00022-7
. K.B. Anderson, J.C. Crelling, Amber, Resinite, and Fossil Resins, ACS Symposium Series, vol. 617 (USA, Am. Chem. Soc., 1996) pp. ix–xvii.
https://doi.org/10.1021/bk-1995-0617
. A. Ross, Amber (Harvard University Press, 1998).
https://archive.org/details/amber00ross/mode/2up
. G. Truic?, N. Ditaranto, M. Caggiani, A. Mangone, S. Li?escu, E. Teodor, L. Sabbatini, G. Radu, Chem. Pap. 68, 15, (2014).
https://doi.org/10.2478/s11696-013-0415-8
. P. Drzewicz, L. Natkaniec-Nowak, D. Czapla, Trends Analyt. Chem. 85, 75 (2016).
https://doi.org/10.1016/j.trac.2016.06.022
. G.I. Truic?, E.D. Teodor, E.S. Teodor, S.C. Li?escu, G.L. Radu, J. Archaeol. Sci. 39, 3524 (2012).
https://doi.org/10.1016/j.jas.2012.05.031
. J.H. Langenheim, C.W. Beck, Science 149, 52 (1965).
https://doi.org/10.1126/science.149.3679.52
. P. Vandenabeele, D.M. Grimaldi, H.G.M. Edwards, L. Moens, Spectrochim. Acta A Mol. Biomol. Spectrosc. 59, 2221 (2003).
https://doi.org/https://doi.org/10.1016/S1386-1425(03)00066-0
. F. Riquelme, P. Northrup, J.L. Ruvalcaba-Sil, V. Stojanoff, D.P. Siddons, J. Alvarado-Ortega, Appl. Phys. A 116, 97 (2014)
https://doi.org/10.1007/s00339-013-8185-2
. G. López-Morales, R. Espinosa Luna, C. Frausto Reyes, Rev. Mex. Fis. 60, 217 (2014).
https://rmf.smf.mx/ojs/index.php/rmf/article/view/4078
. V.L. Villani, F. Caucia, L. Marinoni, A. Leone, M. Brusoni, R. Groppali, J. Gemmol. 35, 752 (2017).
https://gem-a.com/jog-volume-35-issue-8
. G. Beamson, D. Briggs, High resolution XPS of organic polymers: The Scienta ESCA 300 database (UK, John Wiley & Sons, 1992) 22-32.
. S. Beldar, R. Dolasiya, G. Morde, C.S. Narasimhan, Proc. - IEEE Int. Conf. Dielectr. Liq. 2019, 1 (2019).
https://doi.org/10.1109/ICDL.2019.8796589
. Y.M. Shul’ga, V.I. Rubtsov, S. Lobach, N.G. Spitsyna, E.B. Yagubskii, V.N. Vasilets, Synth. Met. 70, 1381 (1995).
https://doi.org/10.1016/0379-6779(94)02887-5
. E. Barletta, K. Wandelt, J. Non-Cryst. Solids 357, 1473 (2011).
https://doi.org/https://doi.org/10.1016/j.jnoncrysol.2010.12.039
. K.E. Fuller, H.D. Downing, M.R. Querry, “Orthorhombic Sulfur (?S)” (Vol. 3 p. 899-922);
https://doi.org/10.1016/B978-012544415-6.50139-4
and: J. Ashok, P.L.H. Varaprasad, J.R. Birch, “Polyethylene (C2H4)n” (Vol. 2 p. 957-987).
https://doi.org/10.1016/B978-0-08-055630-7.50058-4
In: Handbook of optical Constants of Solids, Ed. J.D. Palik (USA, Academic Press, 1998).
. V.V. Tuchin, Phys. Usp. 40, 495 (1997).
https://doi.org/10.1070/PU1997v040n05ABEH000236
. S.L. Jacques, Phys. Med. Biol. 58, R37 (2013).
Downloads
Published
Issue
Section
License
Copyright (c) 2021 Geraldine Vázquez Bautista, M. Zapata Torres, F. Chalé-Lara , M. Meléndez Lira, E. Hernández Rodriguez, E Valaguez Velazquez

This work is licensed under a Creative Commons Attribution 4.0 International License.
©2026 by the authors; licensee SMCTSM, Mexico. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).





